- Which kinds of radiation did Rutherford discover and named?
- alpha and beta particles
- X-rays
- radioactivity
- Which young scientist was the first one to work on the gold-foil experiment?
- Rutherford
- Geiger
- Marsden
- Which substance issued alpha particles in the gold foil experiment?
- Polonium
- Uranium 235
- Radium
- Which fact surprised Rutherford a lot?
- A small number of alpha particles were deflected of an angle larger than 30°
- A few number of alpha particles were reflected by the gold foil and came back
- Most alpha particles went straight on through the gold foil
- What conclusion did Rutherford draw from the experiment?
- The experiment confirmed Thomson's atomic model.
- Electrons travel very fast on their atomic orbits and they can reflect alpha particles in a collision, because of their great energy.
- The positive charges into an atom are all concentrated in a very small region.
- If the alpha particles were slowed down, would more be deflected at greater angles since the nucleus should now more easily turn them back?
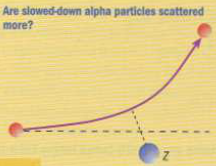
- Yes.
- No.
- Would nuclei of smaller electric charge, as aluminium, scatter alpha particles less strongly, as expected?
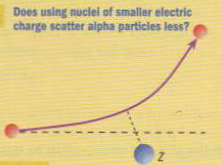
- Yes, they do because electric force is weaker
- No, they do not always because the scattering depend also on the mass
- Would the pattern of numbers of alpha particles scattered at different angles fit the pattern expected from an inverse square law for electrical repulsion?
- Yes.
- No.